| General Information | |
|---|---|
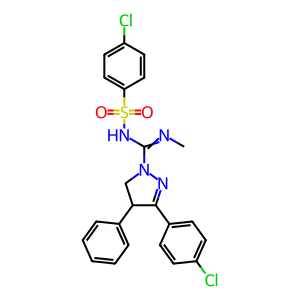
| ZINC ID | ibipinabant |
| Molecular Weight (Da) | 486 |
| SMILES | CN=C(N1N=C([C@H](C1)c1ccccc1)c1ccc(cc1)Cl)NS(=O)(=O)c1ccc(cc1)Cl |
| Molecular Formula | C23H20Cl2N4O2S |
| Action | Antagonist |
| Physicochemical Details | |
|---|---|
| Molar Refractivity | 135.95 |
| HBA | 6 |
| HBD | 1 |
| Rotatable Bonds | 6 |
| Heavy Atoms | 32 |
| LogP | 5.08 |
| Activity (Ki) in nM | 7943.282 |
| Polar Surface Area (PSA) | 82.51 |

| Pharmacokinetic Properties (AdmetSAR) | |
|---|---|
| Human intestinal absorption | + |
| Caco-2 | - |
| Blood brain barrier | + |
| P-glycoprotein inhibitior | + |
| P-glycoprotein substrate | - |
| Cyp3a4 substrate | + |
| Cyp2c9 substrate | - |
| Cyp2d6 substrate | - |
| Cyp3a4 inhibition | - |
| Cyp2c9 inhibition | - |
| Cyp2c19 inhibition | - |
| Cyp2d6 inhibition | - |
| Cyp1a2 inhibition | - |
| Acute oral toxicity | - |
| Carcinogenicity (binary) | - |
| Ames mutagenesis | - |
| Human ether-a-go-go-related gene inhibition | + |
| Biodegradation | 0 |
| Glucocorticoid receptor binding | + |
| Thyroid receptor binding | + |
| Androgen receptor binding | + |
| Plasma protein binding | 0.90082645 |

| Pharmacokinetic Properties (SwissADME) | |
|---|---|
| Number of aromatic heavy atoms | 18 |
| Fraction csp3 | 0.13 |
| Ilogp | 3.31 |
| Xlogp3 | 5.26 |
| Wlogp | 5.08 |
| Mlogp | 4.29 |
| Silicos-it log p | 4.58 |
| Consensus log p | 4.5 |
| Esol log s | -6.2 |
| Esol solubility (mg/ml) | 3.10E-04 |
| Esol solubility (mol/l) | 6.37E-07 |
| Esol class | Poorly sol |
| Ali log s | -6.74 |
| Ali solubility (mg/ml) | 8.82E-05 |
| Ali solubility (mol/l) | 1.81E-07 |
| Ali class | Poorly sol |
| Silicos-it logsw | -8.93 |
| Silicos-it solubility (mg/ml) | 5.79E-07 |
| Silicos-it solubility (mol/l) | 1.19E-09 |
| Silicos-it class | Poorly soluble |
| Pgp substrate | |
| Log kp (cm/s) | -5.54 |
| Lipinski number of violations | 1 |
| Ghose number of violations | 2 |
| Veber number of violations | 0 |
| Egan number of violations | 0 |
| Muegge number of violations | 1 |
| Bioavailability score | 0.55 |
| Pains number of alerts | 0 |
| Brenk number of alerts | 2 |
| Leadlikeness number of violations | 2 |
| Synthetic accessibility | 4.39 |

| Pharmacokinetic Properties (ADMETLab) | |
|---|---|
| Logs | -4.621 |
| Logd | 2.584 |
| Logp | 4.674 |
| F (20%) | 0.001 |
| F (30%) | 0.001 |
| Mdck | 2.43E-05 |
| Ppb | 98.87% |
| Vdss | 0.947 |
| Fu | 2.90% |
| Cyp1a2-inh | 0.323 |
| Cyp1a2-sub | 0.92 |
| Cyp2c19-inh | 0.933 |
| Cyp2c19-sub | 0.934 |
| Cl | 0.535 |
| T12 | 0.035 |
| H-ht | 0.805 |
| Dili | 0.988 |
| Roa | 0.416 |
| Fdamdd | 0.759 |
| Skinsen | 0.056 |
| Ec | 0.003 |
| Ei | 0.006 |
| Respiratory | 0.89 |
| Bcf | 1.062 |
| Igc50 | 4.901 |
| Lc50 | 5.546 |
| Lc50dm | 4.95 |
| Nr-ar | 0.004 |
| Nr-ar-lbd | 0.125 |
| Nr-ahr | 0.28 |
| Nr-aromatase | 0.085 |
| Nr-er | 0.823 |
| Nr-er-lbd | 0.015 |
| Nr-ppar-gamma | 0.49 |
| Sr-are | 0.515 |
| Sr-atad5 | 0.006 |
| Sr-hse | 0.005 |
| Sr-mmp | 0.955 |
| Sr-p53 | 0.402 |
| Vol | 453.636 |
| Dense | 1.071 |
| Flex | 0.231 |
| Nstereo | 1 |
| Nongenotoxic carcinogenicity | 1 |
| Ld50 oral | 0 |
| Genotoxic carcinogenicity mutagenicity | 0 |
| Surechembl | 0 |
| Nonbiodegradable | 1 |
| Skin sensitization | 1 |
| Acute aquatic toxicity | 1 |
| Toxicophores | 1 |
| Qed | 0.426 |
| Synth | 3.008 |
| Fsp3 | 0.13 |
| Mce-18 | 75.692 |
| Natural product-likeness | -0.786 |
| Alarm nmr | 1 |
| Bms | 0 |
| Chelating | 0 |
| Pfizer | 1 |
| Gsk | Rejected |
| Goldentriangle | Accepted |